In a significant advancement for the field of neurodegenerative research, a team of scientists from the Keck School of Medicine of USC’s Mark and Mary Stevens Neuroimaging and Informatics Institute (Stevens INI) has established a new brain imaging benchmark designed to refine the classification of biological changes associated with Alzheimer’s disease (AD). The study, recently published in the journal Imaging Neuroscience, introduces a sophisticated "tau cut-point" that provides a clearer diagnostic lens for Hispanic and non-Hispanic White populations, while simultaneously highlighting critical gaps in how the disease manifests in non-Hispanic Black individuals. This research is a cornerstone of the Health and Aging Brain Study-Health Disparities (HABS-HD), a massive multi-university collaboration led by the University of North Texas Health Science Center and funded by the National Institute on Aging (NIA).
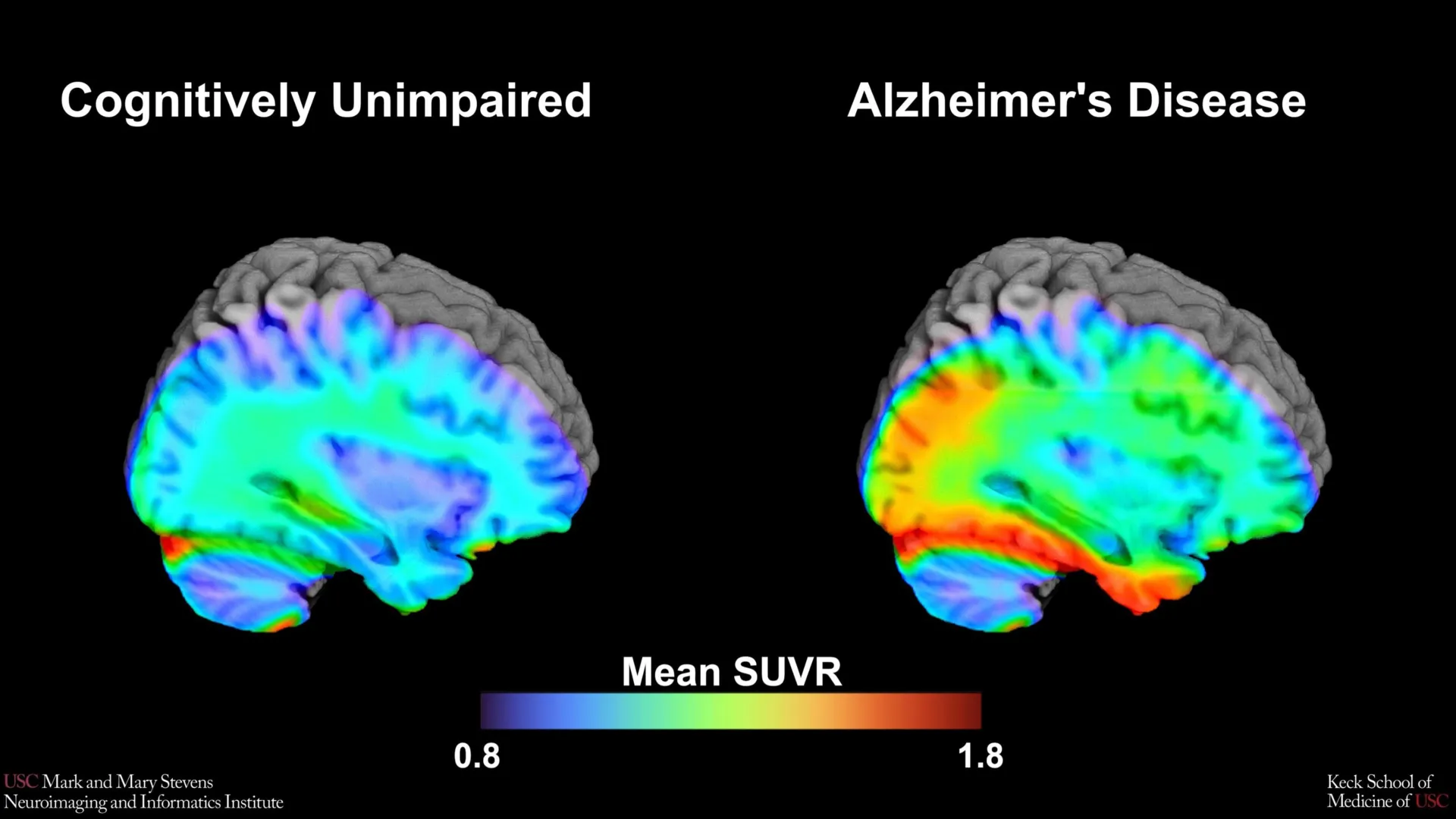
The discovery centers on the use of an advanced positron emission tomography (PET) scan tracer known as 18F-PI-2620. By analyzing the brain scans of over 675 older adults, the USC team sought to identify the specific threshold of tau protein accumulation that distinguishes a healthy aging brain from one showing the early, clinically relevant markers of Alzheimer’s. While the study successfully identified a reliable benchmark for certain demographics, its failure to accurately predict cognitive impairment in Black participants underscores a growing realization in the medical community: Alzheimer’s disease is not a monolithic condition, and its biological drivers can vary significantly across different racial and ethnic groups.
The Biological Mechanics of Alzheimer’s: Amyloid vs. Tau
To understand the importance of this new benchmark, one must first look at the underlying pathology of Alzheimer’s disease. For decades, the "amyloid cascade hypothesis" dominated the field, suggesting that the buildup of amyloid-beta plaques was the primary driver of the disease. However, recent years have seen a shift in focus toward tau protein. While amyloid plaques often appear years before symptoms manifest, the formation of neurofibrillary tangles composed of tau protein is much more closely correlated with actual cognitive decline and the death of neurons.
Tau PET imaging allows researchers to visualize these abnormal proteins in a living brain. By injecting a small amount of a radioactive tracer—in this case, 18F-PI-2620—clinicians can see exactly where tau has accumulated. The tracer binds to the tau tangles, lighting up on the PET scan. The "cut-point" established by the USC researchers refers to the specific level of "tau positivity"—the numerical threshold at which the amount of protein present is high enough to suggest that cognitive impairment is likely caused by Alzheimer’s rather than normal aging.
Methodology and the Role of the HABS-HD Study
The study utilized data from the Health and Aging Brain Study-Health Disparities (HABS-HD), which is currently the most comprehensive longitudinal study of Alzheimer’s in diverse communities. The HABS-HD framework was specifically designed to address the historical lack of diversity in clinical trials, where participants have traditionally been overwhelmingly non-Hispanic White.
The research team, led by senior author Meredith N. Braskie, PhD, and lead author Victoria R. Tennant, analyzed 675 participants who underwent both cognitive testing and neuroimaging. The participants were categorized based on their cognitive status—ranging from cognitively normal to impaired—and their amyloid status. The researchers focused their analysis on the medial temporal lobe, a region deep within the brain that is essential for memory formation and is typically the first area to show tau accumulation in the early stages of Alzheimer’s.
By comparing the PET scans of impaired individuals with those of healthy controls, the team was able to determine that when tau levels in the medial temporal lobe exceeded a specific threshold, it served as a powerful indicator of Alzheimer-related cognitive impairment. However, the data revealed a complex caveat: this indicator was only reliable when the patient also tested positive for amyloid plaques.
The Challenge of Diversity: Disparities in Tau Correlation
Perhaps the most striking finding of the USC study was the discrepancy in how the tau cut-point performed across different racial groups. While the benchmark was highly effective for Hispanic and non-Hispanic White participants, it did not perform as expected for non-Hispanic Black participants. In this group, the presence of tau at the established cut-point did not consistently correlate with cognitive impairment.
"In non-Hispanic Black participants, the tau cut-point did not perform as expected," explained Dr. Meredith Braskie, assistant professor of neurology at the Keck School of Medicine. "This suggests that other pathologies or conditions may be driving cognitive decline in this group."
This finding suggests that for Black populations, cognitive decline may be influenced by a more complex interplay of factors. Researchers speculate that vascular issues, such as small vessel disease, or other proteinopathies like TDP-43, might play a more prominent role. Furthermore, social determinants of health—including lifelong access to quality healthcare, environmental stressors, and systemic inequities—may contribute to different biological manifestations of dementia.
Lead author Victoria R. Tennant, a PhD candidate in USC’s Neuroscience Graduate Program, emphasized that these findings highlight the urgent need for more inclusive research. "The limited reliability of tau as an indicator of cognitive impairment in non-Hispanic Black participants highlights the need for more diverse populations in research and for future studies to examine both biological and social determinants of Alzheimer’s disease," Tennant stated.
A Timeline of Progress in Neuroimaging
The development of the 18F-PI-2620 tracer represents a significant step in the chronology of Alzheimer’s diagnostics.
- Pre-2000s: Alzheimer’s could only be definitively diagnosed through a post-mortem autopsy to identify plaques and tangles.
- 2004: The first amyloid PET tracer (Pittsburgh Compound B) was developed, allowing researchers to see plaques in living patients.
- 2013: The first-generation tau PET tracers began appearing in clinical research, though they often suffered from "off-target binding," where the tracer would stick to areas of the brain that did not actually contain tau.
- 2020-Present: Second-generation tracers like 18F-PI-2620 were introduced. These tracers offer higher specificity and less off-target binding, allowing for the creation of precise "cut-points" like the one identified in this USC study.
This evolution from "clinical observation" to "molecular precision" is transforming how doctors approach the disease, moving the field closer to a "precision medicine" model where treatments are tailored to the individual’s specific biological profile.
Expert Reactions and Institutional Impact
The Mark and Mary Stevens Neuroimaging and Informatics Institute (Stevens INI) has long been at the forefront of mapping the human brain. Director Arthur W. Toga, PhD, noted that this study is part of a broader mission to ensure that no community is left behind as medical technology advances.
"This type of imaging is critical for understanding who is at risk and how the disease develops," said Dr. Toga. "HABS-HD has already produced key findings related to ethnic variations in AD biomarkers, the influences of social determinants on cognitive health, and vascular contributions to dementia. We hope this work will lead to more personalized care and better outcomes for all communities."
The study’s collaborative nature—involving experts from the University of North Texas, the University of Wisconsin-Madison, Washington University in St. Louis, and UC San Francisco—reflects the scale of the challenge. Alzheimer’s is currently the seventh leading cause of death in the United States, and with an aging population, the number of cases is expected to skyrocket by 2050.
Broader Implications for Clinical Trials and Treatment
The establishment of a tau cut-point has immediate implications for the design of clinical trials. Currently, many new Alzheimer’s drugs, such as those targeting the removal of amyloid or tau, require participants to have a specific "biological load" of the disease to be eligible for the trial. Without accurate cut-points, researchers might enroll people who are unlikely to benefit from the drug or exclude those who might.
By providing a validated benchmark for Hispanic and White populations, the USC study allows for more accurate screening of trial participants. However, the study also serves as a warning to the pharmaceutical industry: a "one-size-fits-all" biomarker threshold may inadvertently exclude Black patients or lead to inaccurate conclusions about a drug’s efficacy in that population.
Furthermore, the study reinforces the necessity of the "ATN" framework (Amyloid, Tau, and Neurodegeneration). The fact that the tau cut-point was most effective when amyloid was also present suggests that these biomarkers must be interpreted in tandem rather than in isolation.
Future Directions: Beyond the Biological
As the research team moves forward, the focus will likely shift toward identifying what does drive cognitive decline in populations where tau is not the primary indicator. This will involve integrating neuroimaging data with genetic sequencing, blood-based biomarkers, and detailed assessments of social determinants of health.
The goal is to create a diagnostic toolkit that is as diverse as the population it serves. If Alzheimer’s manifests differently in a Black patient due to vascular contributions or different inflammatory responses, the medical community needs specific benchmarks for those markers as well.
This study by the Keck School of Medicine marks a pivotal moment in the transition toward equitable neurodiagnostics. While it provides a powerful new tool for identifying Alzheimer’s risk in many, it simultaneously shines a light on the work that remains to be done to ensure that every patient, regardless of their background, can receive an accurate and timely diagnosis. By refining these benchmarks, researchers are not just mapping the brain; they are mapping a path toward a future where Alzheimer’s can be predicted, managed, and eventually cured for everyone.