A research team from the Keck School of Medicine of USC’s Mark and Mary Stevens Neuroimaging and Informatics Institute (Stevens INI) has established a significant new brain imaging benchmark that could revolutionize the classification of biologically meaningful changes associated with Alzheimer’s disease (AD). The study, which appears in the journal Imaging Neuroscience, identifies specific "tau cut-points"—thresholds of protein accumulation in the brain—that help distinguish early-stage Alzheimer’s from normal aging. Crucially, the research highlights how these benchmarks perform across different ethnic groups, offering a more nuanced understanding of the disease in Hispanic and non-Hispanic White populations while raising critical questions regarding diagnostic accuracy in non-Hispanic Black communities.
The study was conducted as a central component of the Health and Aging Brain Study-Health Disparities (HABS-HD). This massive, multi-university collaboration, led by the University of North Texas Health Science Center and supported by the National Institute on Aging (NIA), represents one of the most comprehensive efforts to date to understand how Alzheimer’s affects diverse populations. By analyzing 675 older adults, the USC team sought to refine the use of Positron Emission Tomography (PET) scans, specifically focusing on the tau protein, which is increasingly recognized as a more accurate predictor of cognitive decline than the more commonly studied amyloid protein.
The Pathological Landscape: Understanding Amyloid and Tau
To understand the significance of the USC study, it is necessary to examine the biological hallmarks of Alzheimer’s disease. For decades, research focused primarily on amyloid-beta, a protein that forms plaques between neurons. However, while amyloid appears early in the disease progression—sometimes decades before symptoms emerge—it is not always a reliable indicator of imminent cognitive failure. Many individuals carry significant amyloid loads without showing signs of dementia.
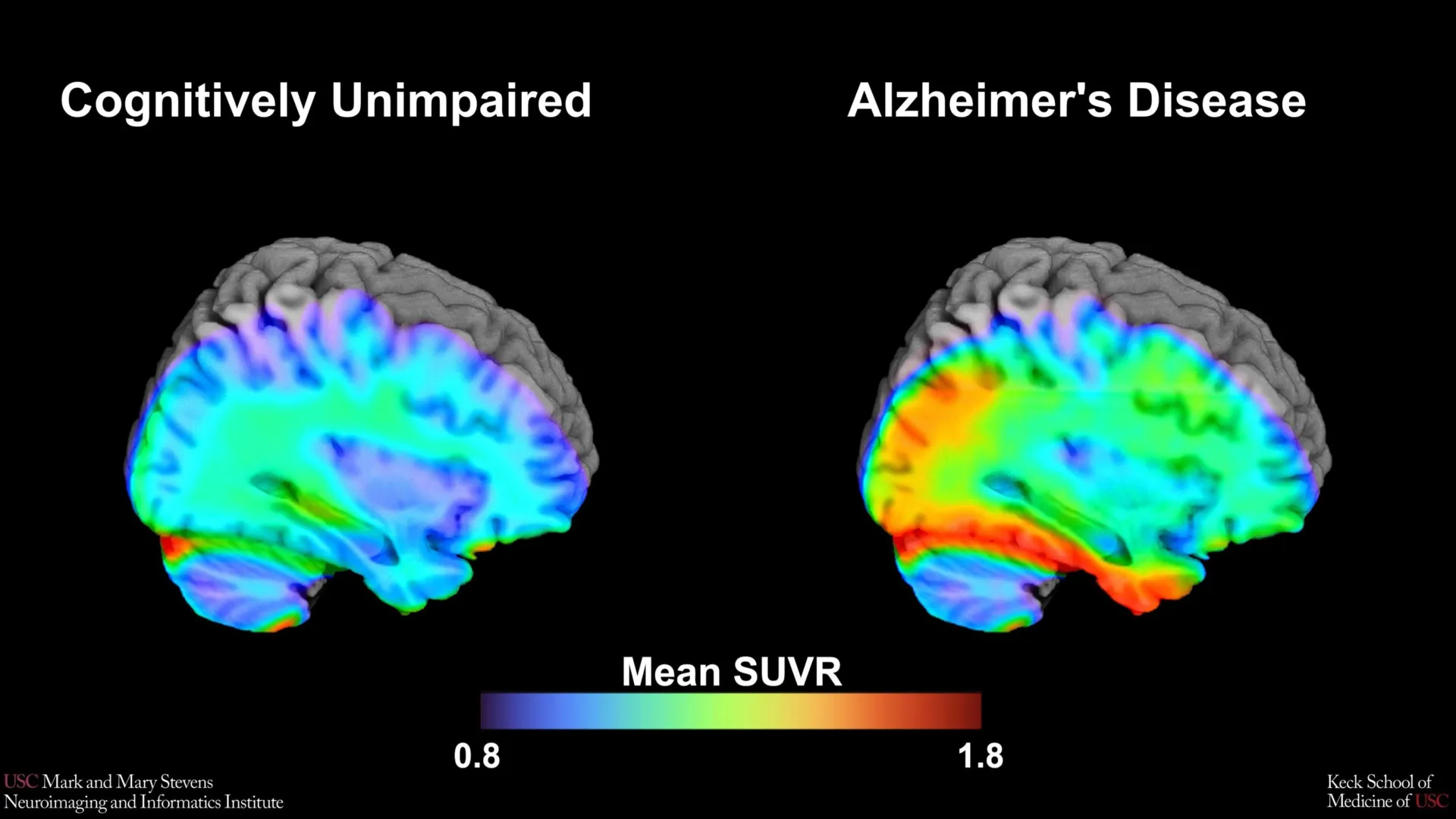
In contrast, the tau protein forms "tangles" inside the neurons themselves. The accumulation of tau is more closely synchronized with the onset of memory loss, disorientation, and the loss of executive function. As tau spreads through the brain, it disrupts cellular transport and eventually leads to cell death. The ability to accurately measure tau via PET scans has transformed Alzheimer’s research from a field reliant on post-mortem analysis to one capable of real-time biological tracking.
The USC researchers utilized an advanced imaging tracer known as 18F-PI-2620. This tracer is designed to bind specifically to tau aggregates, allowing clinicians to visualize the density and location of the protein. By establishing "cut-points," or specific numerical thresholds of tau density, the researchers aimed to create a standardized metric for determining when a patient has crossed the line from healthy aging into the biological territory of Alzheimer’s.
Methodology and Study Design
The HABS-HD study provided a unique dataset, allowing the research team to move beyond the narrow demographic confines of traditional clinical trials, which have historically been dominated by non-Hispanic White participants. The 675 participants were categorized based on cognitive testing into "impaired" and "non-impaired" groups.
The research team focused their analysis on the medial temporal lobe, a region deep within the brain that is essential for memory formation and is typically one of the first areas affected by tau pathology. By comparing the PET scans of cognitively impaired individuals against those with normal cognitive function, the team worked to identify the specific level of 18F-PI-2620 binding that most accurately predicted impairment.
This methodology represents a shift toward "biomarker-based" diagnosis. Rather than waiting for a patient to fail a memory test, researchers are looking for the biological signatures that precede or accompany those failures. The goal is to catch the disease in its prodromal (early) stages, where medical interventions and lifestyle changes might be most effective.
Disparities in Diagnostic Reliability
The most striking finding of the study was the variation in how well the tau cut-points performed across different racial and ethnic groups. For Hispanic and non-Hispanic White participants, the tau benchmark was a highly effective tool for identifying cognitive impairment, provided that another abnormal protein, amyloid, was also present. This "amyloid-plus-tau" model aligns with the current scientific consensus known as the ATN framework (Amyloid, Tau, and Neurodegeneration), which suggests that Alzheimer’s is a multi-step process where amyloid triggers the spread of tau.
However, the results for non-Hispanic Black participants told a different story. In this group, the established tau cut-point did not accurately distinguish between those with and without cognitive impairment. This discrepancy suggests that the biological drivers of dementia in Black populations may involve factors other than, or in addition to, traditional tau and amyloid accumulation.
"In non-Hispanic Black participants, the tau cut-point did not perform as expected," noted senior author Meredith N. Braskie, PhD, an assistant professor of neurology at the Keck School of Medicine. "This suggests that other pathologies or conditions may be driving cognitive decline in this group. Our study is an important step toward better understanding how tau relates to cognition in diverse populations and has important implications for future clinical trials."
Possible alternative drivers of cognitive decline in Black populations include vascular issues—such as hypertension and small vessel disease—as well as the impact of chronic stress and social determinants of health. This finding underscores the danger of a "one-size-fits-all" approach to Alzheimer’s diagnostics and emphasizes the need for a more personalized, inclusive medical model.
Expert Analysis and Official Responses
The leadership at the Stevens Neuroimaging and Informatics Institute emphasized that these findings are part of a broader shift in neuroscientific research. Arthur W. Toga, PhD, director of the Stevens INI, highlighted the importance of the HABS-HD study in addressing long-standing gaps in medical knowledge.
"This type of imaging is critical for understanding who is at risk and how the disease develops," Dr. Toga said. "HABS-HD has already produced key findings related to ethnic variations in AD biomarkers, the influences of social determinants on cognitive health, and vascular contributions to dementia. We hope this work will lead to more personalized care and better outcomes for all communities."
Lead author Victoria R. Tennant, a PhD candidate in USC’s Neuroscience Graduate Program, pointed out that while the study validates the importance of the medial temporal lobe in Alzheimer’s, it also serves as a call to action for the scientific community. She noted that the limited reliability of tau as an indicator in Black participants highlights the urgent need for research that incorporates biological and social determinants of health.
The study’s findings are expected to influence how future clinical trials are designed. If a drug is designed to clear tau from the brain, but tau is not the primary driver of cognitive decline in certain populations, those populations may not benefit from the treatment. Establishing population-specific benchmarks ensures that the right patients receive the right therapies at the right time.
Chronology of Progress in Alzheimer’s Biomarkers
The identification of these tau cut-points is the latest milestone in a decades-long timeline of Alzheimer’s research:
- 1906: Dr. Alois Alzheimer first describes the plaques and tangles in the brain of a deceased patient.
- 1980s-1990s: Researchers identify amyloid-beta and tau as the primary components of these plaques and tangles.
- 2004: The first amyloid PET tracer, Pittsburgh Compound B (PiB), is developed, allowing for the visualization of plaques in living patients.
- 2013: The first tau-specific PET tracers are introduced in research settings.
- 2018: The National Institute on Aging and the Alzheimer’s Association (NIA-AA) propose the ATN framework, shifting the definition of Alzheimer’s from a clinical syndrome to a biological process.
- 2020-Present: Large-scale studies like HABS-HD begin to investigate how these biomarkers manifest in diverse, non-white populations, leading to the USC team’s current findings on 18F-PI-2620.
Implications for the Future of Alzheimer’s Care
The establishment of a tau cut-point using the 18F-PI-2620 tracer provides clinicians with a concrete "line in the sand." In a clinical setting, this could mean that a doctor looking at a PET scan can now say with greater statistical confidence whether a patient’s tau levels are indicative of early Alzheimer’s or are simply part of the normal aging process for their demographic.
Furthermore, this research supports the move toward "precision medicine." By acknowledging that Alzheimer’s may look different in the brain of a Hispanic patient versus a Black patient, the medical community can move away from generalized treatments. For instance, if vascular health plays a larger role in cognitive decline for Black patients, a treatment plan might focus more heavily on blood pressure management and cardiovascular health alongside traditional Alzheimer’s medications.
The study also highlights the necessity of the "amyloid-first" hypothesis in certain groups. Because the tau cut-point was only effective when amyloid was also present, it reinforces the idea that tau-targeting therapies might be most effective when administered to patients who have already tested positive for amyloid.
Conclusion
The work of the USC Stevens INI team represents a vital step toward equity in neurological healthcare. As the global population ages, the prevalence of Alzheimer’s is expected to rise, with a disproportionate impact on minority communities. By refining the tools used to diagnose the disease and ensuring they are validated across diverse populations, researchers are laying the groundwork for a future where Alzheimer’s can be managed, or perhaps even prevented, for everyone.
The study involved a massive collaborative effort, including authors from the Alzheimer’s Therapeutic Research Institute at USC, the University of Wisconsin-Madison, Washington University in St. Louis, and the University of California, San Francisco. This cross-institutional approach reflects the complexity of the disease and the collective resolve required to solve it. As research continues under the HABS-HD umbrella, the medical community inches closer to a comprehensive understanding of the brain’s most daunting challenge.