In a significant advancement for neurodegenerative research, a multidisciplinary team from the Keck School of Medicine of USC’s Mark and Mary Stevens Neuroimaging and Informatics Institute (Stevens INI) has established a new brain imaging benchmark. This benchmark is designed to enhance the classification of biologically meaningful changes associated with Alzheimer’s disease (AD), specifically targeting the nuances found within Hispanic and non-Hispanic White populations. The study, recently published in the journal Imaging Neuroscience, represents a pivotal component of the Health and Aging Brain Study-Health Disparities (HABS-HD), a comprehensive multi-university collaboration led by the University of North Texas Health Science Center and supported by the National Institute on Aging (NIA).
The identification of this benchmark addresses a long-standing challenge in neurology: the need for precise, objective measures to distinguish between normal age-related cognitive decline and the early pathological stages of Alzheimer’s disease. By focusing on tau protein accumulation—a primary hallmark of AD—the researchers have provided a framework that could revolutionize clinical diagnostics and the selection process for clinical trials.
The Evolution of Alzheimer’s Diagnostic Frameworks
For decades, the diagnosis of Alzheimer’s disease relied heavily on clinical symptoms and post-mortem examinations. However, the advent of Positron Emission Tomography (PET) imaging has shifted the paradigm toward biological markers. Traditionally, amyloid-beta plaques were the primary focus of AD research. While amyloid buildup is an early indicator of the disease, it often lacks a direct correlation with the severity of cognitive impairment.
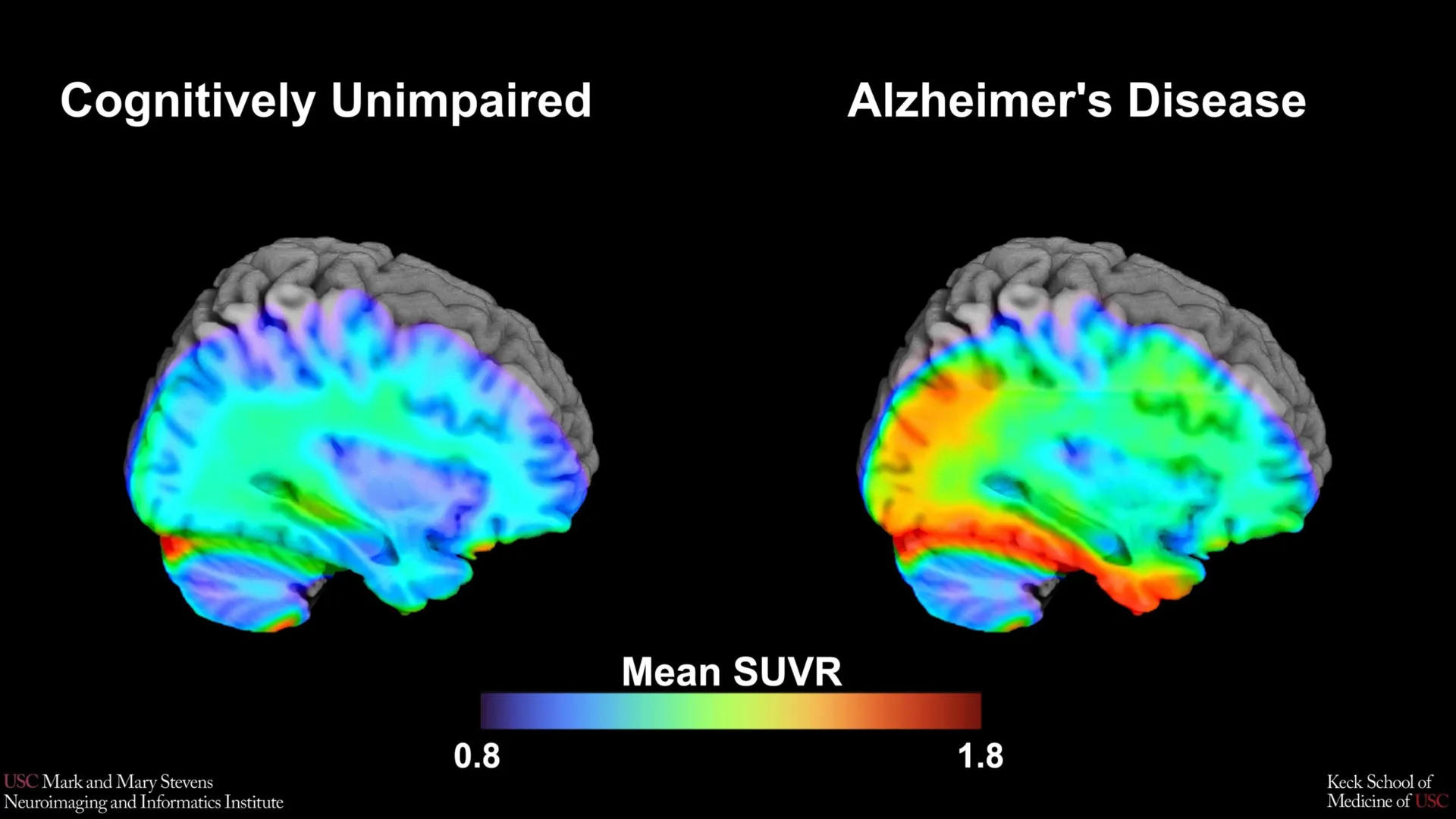
In contrast, tau protein tangles—neurofibrillary structures that disrupt nutrient transport within neurons—are more closely linked to the actual manifestation of symptoms such as memory loss and executive dysfunction. The USC study leverages this relationship by utilizing tau PET scans to establish "cut-points," which are specific thresholds of protein accumulation that indicate a transition from healthy aging to a disease state.
The research team analyzed data from over 675 older adults participating in the HABS-HD study. This cohort is unique due to its emphasis on diversity, addressing a historical gap in medical literature where minority populations were often underrepresented. The objective was to identify the optimal brain signal that distinguishes individuals with clinically relevant biological markers of AD from those who are aging without significant pathology.
Methodology and the Role of 18F-PI-2620
Central to the study’s methodology was the use of an advanced imaging tracer known as 18F-PI-2620. Unlike earlier generations of tau tracers, 18F-PI-2620 offers high affinity and selectivity for the tau aggregates specifically associated with Alzheimer’s disease, reducing "off-target" binding that can sometimes obscure results in other regions of the brain.
The researchers focused their analysis on the medial temporal lobe, a region deep within the brain that is critical for memory formation and is typically one of the first areas affected by tau pathology. By measuring the "Standardized Uptake Value Ratio" (SUVR) of the tracer in this region, the team was able to calculate a specific cut-point.
The process involved comparing the scans of cognitively impaired participants—determined through rigorous neuropsychological testing—with those of cognitively normal individuals. The goal was to find a numerical value of tau density that served as a reliable "red flag" for Alzheimer’s-related impairment.
Findings Across Racial and Ethnic Demographics
The study’s findings revealed a complex landscape regarding the universality of AD biomarkers. The established tau cut-point demonstrated high efficacy in distinguishing cognitive impairment among Hispanic and non-Hispanic White participants. However, this effectiveness was contingent upon the presence of amyloid-beta. The researchers found that the tau benchmark was most predictive when "amyloid-positive" status was also confirmed, supporting the "amyloid cascade hypothesis," which suggests that amyloid facilitates the spread of tau throughout the cortex.
Surprisingly, the benchmark did not perform with the same level of reliability in non-Hispanic Black participants. In this group, the tau cut-point did not align as expected with cognitive impairment levels. This discrepancy suggests that the drivers of cognitive decline in Black populations may involve a different constellation of pathologies.
"Our tau cut-point was able to distinguish whether study participants had cognitive impairment—but only when another abnormal protein, amyloid, was also present in those with cognitive impairment, and only in Hispanic and non-Hispanic White participants," noted senior author Meredith N. Braskie, PhD, an assistant professor of neurology at USC. "In non-Hispanic Black participants, the tau cut-point did not perform as expected. This suggests that other pathologies or conditions may be driving cognitive decline in this group."
Addressing the Data Gap in Minority Health
The failure of the tau benchmark to translate across all demographic groups highlights a critical area of concern in modern medicine: health disparities. For years, the medical community has recognized that Alzheimer’s disease disproportionately affects Black and Hispanic communities, yet the biological reasons for these disparities remain under-researched.
The HABS-HD study, from which this data was drawn, is designed specifically to bridge this gap. By examining both biological and social determinants of health, the project aims to understand why certain populations may be more susceptible to vascular contributions to dementia or why traditional biomarkers like tau might not be the primary drivers of decline in all groups.
Lead author Victoria R. Tennant, a PhD candidate in USC’s Neuroscience Graduate Program, emphasized the need for inclusivity in research. "The limited reliability of tau as an indicator of cognitive impairment in non-Hispanic Black participants highlights the need for more diverse populations in research and for future studies to examine both biological and social determinants of Alzheimer’s disease," she stated.
The findings suggest that for Black patients, cognitive decline might be more closely related to vascular issues, such as hypertension or small vessel disease, or perhaps different proteinopathies that are not yet as well-characterized as tau and amyloid.
Chronology of the HABS-HD Initiative
The Health and Aging Brain Study-Health Disparities (HABS-HD) has been a multi-year effort to redefine the understanding of dementia.
- Phase 1: Initial recruitment focused on establishing a large, diverse cohort of Mexican American and non-Hispanic White individuals to study the influence of genetics and environment on AD.
- Phase 2: Expansion of the study to include non-Hispanic Black participants, acknowledging the high prevalence of dementia in this community.
- Phase 3: Integration of advanced neuroimaging (Tau and Amyloid PET) and blood-based biomarkers to create a multi-modal diagnostic toolkit.
- Current Findings: The publication in Imaging Neuroscience marks a critical milestone in this timeline, providing the first validated tau cut-points using the 18F-PI-2620 tracer within this specific diverse cohort.
Broader Implications for Clinical Trials and Personalized Medicine
The establishment of a tau cut-point has immediate implications for the design of clinical trials. As pharmaceutical companies develop drugs aimed at clearing tau from the brain, they require precise inclusion criteria to ensure they are testing the medication on the right patients. A standardized cut-point allows researchers to identify "tau-positive" individuals who are in the early, potentially reversible stages of the disease.
Furthermore, these findings advocate for a more personalized approach to neurology. If the biological drivers of Alzheimer’s vary by ethnicity or race, then a "one-size-fits-all" diagnostic or treatment plan is likely to fail large segments of the population.
Arthur W. Toga, PhD, director of the Stevens INI, highlighted the broader impact of the institute’s work. "This type of imaging is critical for understanding who is at risk and how the disease develops," Toga said. "These findings are just the latest to come from HABS-HD, which is the most comprehensive study of Alzheimer’s disease and related dementias in diverse communities. We hope this work will lead to more personalized care and better outcomes for all communities."
The Stevens INI has been at the forefront of this movement, producing data that links ethnic variations in biomarkers to vascular health and social determinants. By refining the tools used to measure the brain, the institute is providing the foundation for a future where Alzheimer’s can be caught and treated long before debilitating symptoms emerge.
Future Research Directions
Following the results of this study, the research team plans to investigate the alternative factors contributing to cognitive impairment in non-Hispanic Black populations. This includes a deeper look into "white matter hyperintensities"—indicators of vascular damage—and the role of inflammation and chronic stress, which are often linked to social determinants of health.
Additionally, the team will continue to monitor the HABS-HD cohort longitudinally. Longitudinal data will allow researchers to see if the tau cut-points identified today can accurately predict the rate of cognitive decline over the next five to ten years.
The study was a massive collaborative effort, involving experts from the Alzheimer’s Therapeutic Research Institute (ATRI), the Waisman Center at the University of Wisconsin-Madison, Washington University School of Medicine, and the University of California, San Francisco. This level of institutional cooperation underscores the urgency and scale of the challenge posed by Alzheimer’s disease in an aging global population.
As the scientific community moves closer to effective therapies, the ability to accurately categorize patients through benchmarks like the USC tau cut-point will be the difference between clinical success and failure. For the millions of families affected by Alzheimer’s, particularly those in underserved communities, this research offers a pathway toward more equitable and effective healthcare.