A team of researchers from the Keck School of Medicine of USC’s Mark and Mary Stevens Neuroimaging and Informatics Institute (Stevens INI) has identified a new brain imaging benchmark that may improve how researchers classify biologically meaningful changes associated with Alzheimer’s disease, especially in Hispanic and non-Hispanic White populations. This groundbreaking study, recently published in the journal Imaging Neuroscience, represents a pivotal advancement in the field of neuroimaging. It is a core component of the Health and Aging Brain Study-Health Disparities (HABS-HD), a multi-university collaboration led by the University of North Texas Health Science Center and supported by the National Institute on Aging. By establishing more precise "cut-points" for tau protein accumulation, the research provides a vital tool for distinguishing normal aging from the early biological signals of Alzheimer’s disease (AD).
Establishing a New Diagnostic Threshold
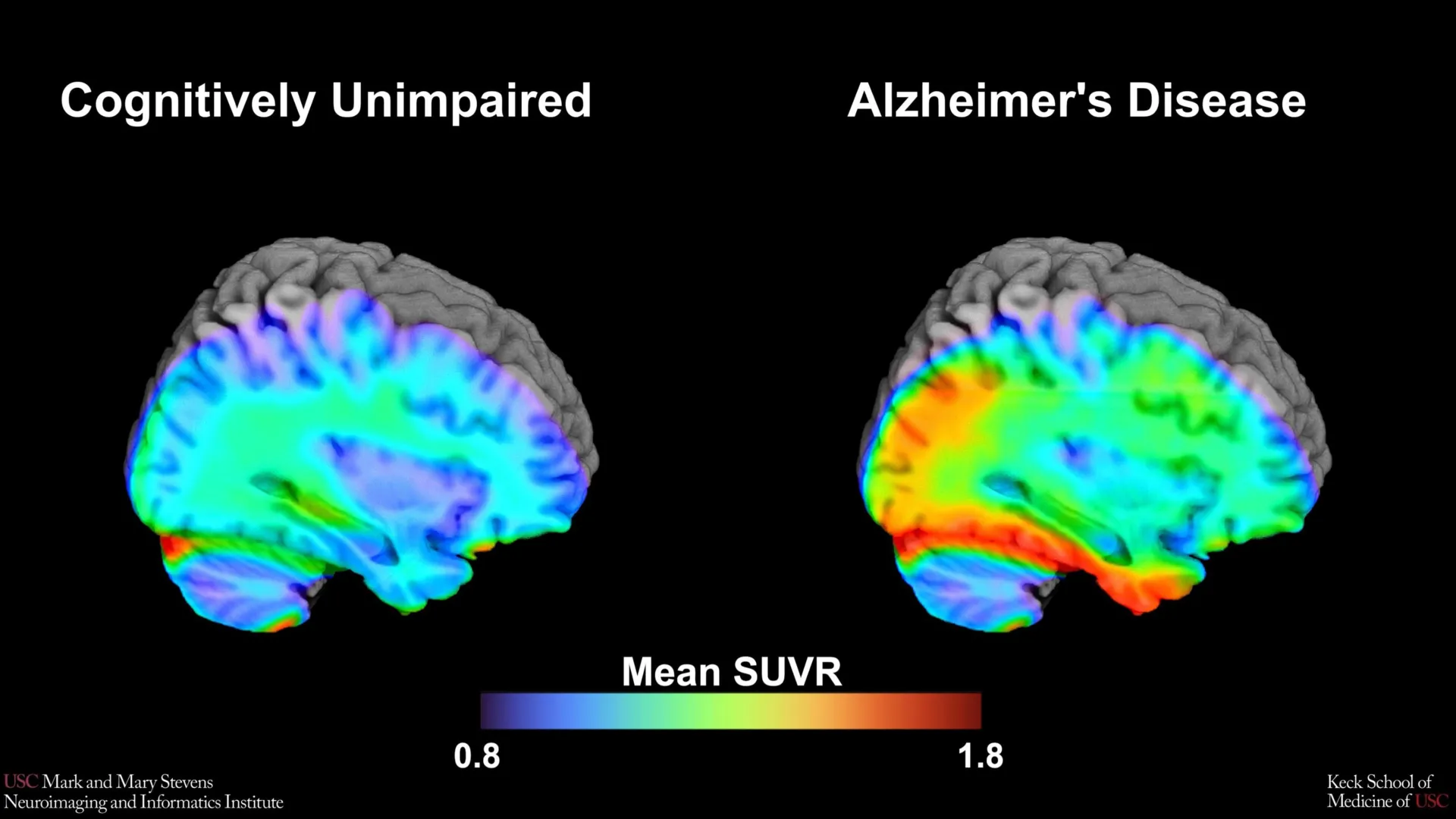
The identification of this new benchmark stems from the need for more objective, quantifiable measures in the diagnosis of neurodegenerative diseases. Traditionally, Alzheimer’s disease has been difficult to diagnose in its earliest stages, as the initial cognitive symptoms often overlap with the natural aging process. The USC-led team utilized advanced Positron Emission Tomography (PET) scans to observe the presence of tau protein, a hallmark of Alzheimer’s pathology that forms "tangles" within the neurons, disrupting cell communication and eventually leading to cell death.
Using a specific imaging tracer known as 18F-PI-2620, the research team analyzed a cohort of over 675 older adults. The goal was to pinpoint the exact level of tau accumulation that serves as a tipping point for clinical cognitive impairment. This threshold, or "cut-point," acts as a biomarker that allows clinicians to categorize patients based on their risk level. According to the study, when tau levels in the medial temporal lobe—a region of the brain critical for memory and emotion—exceeded a specific threshold, it became a strong indicator of cognitive impairment related to Alzheimer’s.
The Role of HABS-HD and the Importance of Diversity
The study’s findings are deeply embedded in the Health and Aging Brain Study-Health Disparities (HABS-HD) framework. HABS-HD is the most comprehensive study of Alzheimer’s disease and related dementias focused specifically on diverse communities. For decades, clinical trials for Alzheimer’s have been criticized for a lack of diversity, often relying on data from predominantly non-Hispanic White populations. This lack of representation has created gaps in the medical community’s understanding of how the disease manifests in different ethnic and racial groups.
The inclusion of Hispanic and non-Hispanic Black participants in this study revealed a complex reality. While the tau cut-point was highly effective at identifying cognitive impairment in Hispanic and non-Hispanic White participants, it did not perform with the same reliability in non-Hispanic Black participants. This discrepancy suggests that cognitive decline in Black populations may be driven by different or additional underlying pathologies, such as vascular issues or social determinants of health, rather than solely by the accumulation of tau protein.
Scientific Context: Tau Versus Amyloid
To understand the significance of this benchmark, it is essential to distinguish between the two primary proteins associated with Alzheimer’s: amyloid-beta and tau. Amyloid-beta typically forms plaques outside of neurons and is often one of the earliest signs of the disease, appearing years or even decades before cognitive symptoms manifest. However, the presence of amyloid alone does not always lead to immediate cognitive decline.
Tau, on the other hand, is more closely correlated with the actual onset of memory loss and functional impairment. As tau tangles spread from the medial temporal lobe to other parts of the cortex, symptoms become more severe. The USC study found that the tau cut-point was most effective when another abnormal protein, amyloid, was also present. This "dual-protein" requirement highlights the synergistic relationship between amyloid and tau in the progression of Alzheimer’s, particularly in Hispanic and White populations.
Chronology of Alzheimer’s Diagnostic Evolution
The journey toward this new benchmark is part of a decades-long evolution in neuroimaging.
- 1980s-1990s: Diagnosis was largely based on clinical observation and cognitive testing, with definitive confirmation only possible through post-mortem brain autopsies.
- Early 2000s: The advent of Amyloid PET imaging allowed researchers to see plaques in living patients for the first time, though it could not accurately predict the timing of cognitive decline.
- 2010s: The development of first-generation tau tracers enabled the visualization of tangles, but these tracers often suffered from "off-target binding," where the tracer would stick to non-tau proteins, muddling the data.
- 2020-Present: Second-generation tracers like 18F-PI-2620, used in the USC study, offer much higher specificity and lower off-target binding. This technological leap has made it possible to establish the precise cut-points reported in the current study.
Technical Insights into the 18F-PI-2620 Tracer
The choice of the 18F-PI-2620 tracer was instrumental to the study’s success. Unlike earlier tracers, 18F-PI-2620 is designed to bind specifically to the "three-repeat" and "four-repeat" tau isoforms associated with Alzheimer’s. This allows for a clearer picture of the protein’s density in the medial temporal lobe.
Lead author Victoria R. Tennant, a PhD candidate in USC’s Neuroscience Graduate Program, emphasized that while the medial temporal lobe has long been a region of interest, the ability to define "tau positivity" using this specific tracer is a major step forward for both research and clinical application. By standardizing what constitutes a "positive" scan, the medical community can move toward a more objective diagnostic framework that relies less on subjective cognitive assessment and more on biological reality.
Official Responses and Expert Analysis
The implications of this research have been met with enthusiasm and a call for further inquiry from the academic community. Arthur W. Toga, PhD, director of the Stevens INI, noted that this type of imaging is critical for understanding who is at risk and how the disease develops. "These findings are just the latest to come from HABS-HD," Toga said. "We hope this work will lead to more personalized care and better outcomes for all communities."
Senior author Meredith N. Braskie, PhD, assistant professor of neurology, highlighted the cautionary aspect of the findings regarding non-Hispanic Black participants. "In non-Hispanic Black participants, the tau cut-point did not perform as expected," Braskie stated. "This suggests that other pathologies or conditions may be driving cognitive decline in this group. Our study is an important step toward better understanding how tau relates to cognition in diverse populations and has important implications for future clinical trials that aim to target tau."
Industry analysts suggest that these findings could force pharmaceutical companies to rethink their inclusion criteria for clinical trials. If tau is not the primary driver of cognitive decline in all populations, then tau-targeting drugs may not be equally effective for everyone.
Broader Impact on Clinical Trials and Personalized Medicine
The establishment of tau cut-points is expected to have a direct impact on the design of future clinical trials. Currently, many trials for Alzheimer’s drugs require participants to have a certain level of amyloid or tau "burden" to be included. By providing a more refined benchmark, the USC study allows for better screening of participants, ensuring that those enrolled are the most likely to benefit from the therapy being tested.
Furthermore, the study underscores the necessity of a "precision medicine" approach to Alzheimer’s. Rather than a one-size-fits-all diagnostic model, clinicians may eventually need to use different benchmarks or look for different biomarkers based on a patient’s ethnic or genetic background. This is particularly important as the global population ages and the prevalence of Alzheimer’s continues to rise.
Data and Statistical Significance
The study’s robust dataset included a wide range of cognitive assessments alongside the PET imaging. Participants were categorized into "cognitively unimpaired" and "cognitively impaired" groups based on standardized neuropsychological testing. The researchers then used statistical modeling to determine which tau levels best predicted a participant’s cognitive status.
The data revealed that in Hispanic and White populations, the medial temporal lobe tau levels were highly predictive of impairment, with a high "area under the curve" (AUC) in statistical tests, signifying strong diagnostic accuracy. However, the lower predictive power in Black participants remains a focal point for future research. Scientists are now looking into whether vascular health—such as hypertension and diabetes, which disproportionately affect Black communities—might play a more significant role in their cognitive health than tau protein accumulation.
Conclusion and Future Directions
The research conducted by the Stevens INI team represents a significant milestone in the quest to demystify Alzheimer’s disease. By identifying a new brain imaging benchmark, the study provides a clearer roadmap for early diagnosis and a more nuanced understanding of how the disease affects different populations.
In addition to Tennant and Braskie, the study involved a massive collaborative effort including researchers from the Alzheimer’s Therapeutic Research Institute at USC, the University of Wisconsin-Madison, Washington University in St. Louis, and the University of California, San Francisco. The multi-institutional nature of the study reflects the complexity of the disease and the necessity of a unified scientific front.
As the HABS-HD initiative continues, the focus will likely shift toward identifying the "missing" biomarkers in populations where tau and amyloid do not tell the whole story. This work is supported by multiple grants from the National Institute on Aging and the National Institutes of Health, ensuring that the search for equity in Alzheimer’s diagnostics remains a national priority. Through such rigorous, inclusive research, the medical community moves closer to a future where personalized, effective care for Alzheimer’s is accessible to all, regardless of ethnicity or background.